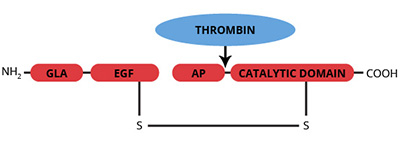
The vitamin K-dependent zymogen, protein C, is synthesized in the liver as a single chain polypeptide and is subsequently converted to a disulfide linked heterodimer, by removal of a dipeptide (Lys-146 and Arg-147) from the precursor molecule (1,2). Trace quantities of the single chain form have been observed in plasma. The light chain, which is responsible for the calcium dependent binding of protein C to phospholipid vesicles, contains 11 γ-carboxyglutamic acid (gla) residues, 1 b-hydroxyaspartic acid residue, and 2 epidermal growth factor (EGF) homology domains. The serine protease catalytic triad is located in the heavy chain. Human protein C is susceptible to proteolytic cleavage of a peptide (Mr=3000) from the COOH-terminal end of the heavy chain, yielding an altered form referred to as β-protein C. No functional distinction between α- and β-protein C has been observed. A single cleavage at Arg-12 (Arg-14 in bovine) of the heavy chain of human protein C converts the zymogen into the serine protease, activated protein C. This cleavage is catalyzed by a complex between α-thrombin and the endothelial cell surface protein thrombomodulin. In contrast to the other vitamin K dependent coagulation factors, activated protein C functions as an anticoagulant by catalyzing the proteolytic inactivation of factors Va and VIIIa. APC also contributes to the fibrinolytic response by complex formation with plasminogen activator inhibitors.
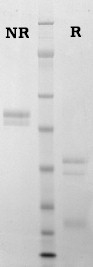
Bovine protein C is prepared from fresh citrated bovine plasma by a modification of the Walker procedure (3), as described by Haley et al. (4). Human protein C is prepared from fresh frozen citrated human plasma using a combination of immunoaffinity chromatography (5), and conventional techniques (4,9). Protein C is provided in 50% (vol/vol) glycerol/H2O and should be stored at -20oC. Purity is determined by SDS-PAGE analysis and activity is measured using a chromogenic substrate based assay.