No overview available.
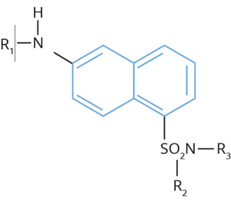
General Structure of ANSN-Based Florogenic Substrates
The general structure of 6-amino-1-naphthalenesulfonamide-based (ANSN) fluorogenic substrates is illustrated. In the present family of compounds, R1 is a tripeptide of which the COOH-terminal residue is typically an arginine. R2 and R3 may be either a hydrogen, alkyl, aryl, or cycloalkyl group. The dotted line indicates the site of hydrolysis by the various serine proteases.
Substrates containing the fluorescent reporter group 6-amino-1-naphthalene-sulfonamide (ANSN) are useful compounds for monitoring the enzyme activity of various serine proteases (1-12). In this class of compounds, the ANSN reporter group is linked (in the R1 position) to short tri-peptide sequences. The peptide sequences are designed to optimize the interaction between the enzyme and substrate. Additional components which may be added to the R2 and R3 positions reflect changes in the P’ subsite positions, and generally affect the kinetic parameters of the substrates. Compounds which are effective substrates are hydrolyzed between the tri-peptide and the ANSN group. Once cleaved from the peptide moiety, the ANSN group exhibits about a 1000 fold increase in relative fluorescence.
The kinetic properties identified on the following page will aid in the selection of an appropriate substrate. The ANSN substrates have proved especially useful for the analyses of factor VIIa (1-5,7,9,10,12). Although the substrate hydrolysis rates are relatively slow for factor VIIa alone, several compounds such as compound SN-17a exhibit a large change in kcat when tissue factor is incorporated into the assay system.
Usage Information:
ANSN-based substrates are provided as 10mM stock solutions in DMSO, and are generally useful for analyzing enzymes within a concentration range of 400 nM to as low as 50pM. ANSN substrates are typically used at final concentrations of 50 to 100 µM and may be diluted to this final concentration in most physiologic buffers (ie., TBS, HBS, PBS, etc). Reactions (the relative change in fluorescence) may be monitored using standard fluorometers or fluorescent plate readers. Kinetic or endpoint modes may be used with the instrument set at the following wavelengths (excitation=352nm, emission=470nm). Light artifacts can be minimized by employing a 390 to 450 nm long-pass cutoff filter in the emission beam.
Kinetic properties of each substrate are listed in the table below, and a list of journal references is included below the table for examples of use.
The stock substrate solutions in DMSO should be stored frozen at 4°C or colder, and should be protected from light. Under these conditions the compounds will remain stable for over one year.
**Key for tables below: Km = µM; kcat = s-1; kcat/Km = M-1s-1 x 10-5 ; ND= Not Determined
No properties available.
- Butenas, S., et al., Biochemistry, 31, 5399 (1992).
- Lawson, J.H., et al., J. Biol. Chem., 267, 4834 (1992).
- Lawson, J.H., et al., Methods in Enzymology, 222, 177 (1993).
- Butenas, S., et al., Biochemistry, 32, 6531 (1993).
- Butenas, S., et al., J. Biol. Chem., 269, 25838 (1994).
- Butenas, S., et al., Anal. Biochem., 225, 231 (1995).
- Butenas, S., and Mann, K.G., Biochemistry, 35, 1904 (1996).
- Butenas, S., et al., Biochemistry, 36, 2123 (1997).
- Butenas, S., et al., J. Biol. Chem., 272, 21527 (1997).
- Butenas, S., et al., Thromb. Haemost., 78, 1193 (1997).
- Hockin,M.F.,et al.,Arterioscler.Thromb.Vasc.Biol.,in press(1997).
- Butenas, S., et al., U.S.A. patent # 5,399,487.
- Komissarov, A., et al., Am J Physiol Lung Cell Mol Physiol (August 7, 2009). (SN-5)
- Butenas et al. Thromb Haemost 1997;78:1193-201.
This publication list is not all encompassing, and is only meant to provide limited examples of how Prolytix products are used. We encourage you to search the literature for other examples pertinent to your experimentation, and to contact us with any technical questions.
